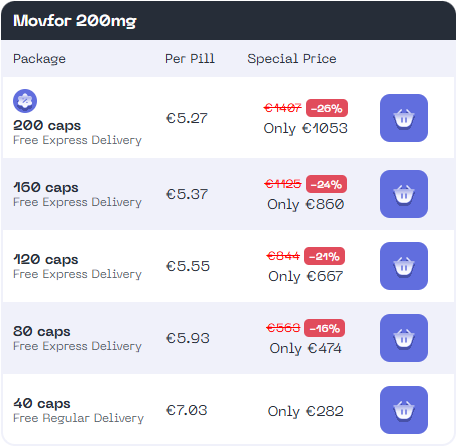
Buy MOVFOR® 200MG Capsules Online
- Brand: MOVFOR®
- Composition: Molnupiravir
- Power: 200mg
- Pack Type: Bottle
- Drug form: Capsules
- Disease/Treatment: experimental drug under study for the treatment of mild to moderate COVID-19
- Manufacturer: Hetero Healtchare Ltd
- Country of Origin: India
Order MOVFOR® 200 MG Molnupiravir Capsules
 MOVFOR® 200 MG is a drug for the treatment of coronavirus infection (COVID-19) of mild or moderate severity in adults, including those with an increased risk of disease progression to severe and not requiring additional oxygen therapy from the Indian pharmaceutical company Hetero Healthchare.
MOVFOR® 200 MG is a drug for the treatment of coronavirus infection (COVID-19) of mild or moderate severity in adults, including those with an increased risk of disease progression to severe and not requiring additional oxygen therapy from the Indian pharmaceutical company Hetero Healthchare.
A new substance has been created, called Molnupiravir in honor of the hammer of the Scandinavian god Thor Mjolnir. Studies, in particular independent trials conducted in India, show that the drug is able to effectively prevent the reproduction of coronavirus, reducing the risk of severe disease and infection of other people.
It is an analog of a ribonucleoside, the particle that makes up the RNA of the virus. The drug is embedded in the genome of the causative agent of the disease, disrupting the process of replication, in other words, reproduction. Due to this, the number of viral particles in the human body does not grow, and they cease to spread throughout the body.
What do we know about its effectiveness MOVFOR® 200 MG Molnupiravir Capsules?
 According to research data, the drug was effective against all the most common COVID-19 strains: delta, gamma and mu. To date, variants with these strains have accounted for almost 80% of the cases evaluated in the study.
According to research data, the drug was effective against all the most common COVID-19 strains: delta, gamma and mu. To date, variants with these strains have accounted for almost 80% of the cases evaluated in the study.
According to the interim analysis of the third phase of studies, the drug reduces the risk of hospitalization and death from coronavirus by half. Only 7.3% of patients receiving Molnupiravir were hospitalized or died. In the placebo group, there were 14.1% of them.
At the same time, it is important to note: the studies conducted give us grounds to assert that the drug improves the course of the disease in patients with mild and moderate course of the disease.
At first, molnupiravir was developed by the pharmaceutical company Drug Innovation Ventures at Emory (DRIVE) for the treatment of influenza and respiratory syncytial virus. But with the beginning of the pandemic, the plans changed. The development was sold to Ridgeback Biotherapeutics, which later signed a contract with Merck, and began to investigate against coronaviruses. At the end of 2020, the compound was successfully tested on ferrets infected with SARS-CoV-2 — and it showed a decrease in the amount of the virus in the respiratory tract.
Scientists assumed that molnupiravir is embedded in the genome of the virus, but the full mechanism of action of the drug has become clear only recently. The coronavirus genome is an RNA molecule. When the virus multiplies, it uses four types of “bricks” for this. Molnupiravir pretends to be one of them and is embedded in the virus. But because he’s not like that, the virus mutates. For some time, the pathogen still continues to multiply, but mutations accumulate and at some point the virus finally dies — in fact, mutates to death.
But remdesivir, which is used in the treatment of severe covid, acts a little differently. As soon as it is embedded in the virus, the reproduction of the pathogen is immediately disrupted. But some untouched copies of the virus still manage to escape. Perhaps that is why many scientists have doubts about the benefits of the drug. WHO said a year ago that it does not recommend the use of remdesivir in patients with COVID-19.
How to take MOVFOR® 200 MG Molnupiravir Capsules?
The drug MOVFOR® 200 MG is taken orally regardless of food intake.
Capsules should be swallowed whole, without opening, crushing or chewing them, washed down with a sufficient amount of liquid (for example, a glass of water).
The use of MOVFOR® 200 MG is possible only under the supervision of a doctor!
What is the dosage for MOVFOR® 200 MG?
- For the treatment of a new coronavirus infection (COVID-19) caused by the SARS-CoV-2 virus, the following dosage regimen is recommended in adults:
– 4 capsules 200 mg or 2 capsules 400 mg orally 2 times a day (every 12 hours). - A single dose is 800 mg.
- The daily dose is 1600 mg. The duration of the course of treatment is 5 days.
Treatment with the drug Molnupiravir MOVFOR® 200 MG should be started as early as possible after the diagnosis of a new coronavirus infection (COVID-19) and / or within 5 days after the appearance of the first symptoms of the disease.
Missed dose
In case of missing the next dose of the drug, if the delay in admission was less than 10 hours from the scheduled time of admission, then the missed dose should be taken as soon as possible and the usual dosing regimen should be resumed; if the delay in admission was more than 10 hours, then the missed dose should not be taken, and the next dose is taken at the usual time.
The patient should not take a double dose of the drug to compensate for the missed dose.
Special patient groups for the drug MOVFOR® 200 MG:
Old age
- Dose adjustment of MOVFOR® 200 MG is not required depending on age.
Kidney failure
- Dose adjustment of MOVFOR® 200 MG is not required for patients with renal insufficiency.
Liver failure
- Dose adjustment of MOVFOR® 200 MG is not required for patients with hepatic insufficiency.
Children
- There is no data on the safety and efficacy of MOVFOR® 200 MG in children under 18 years of age.
What are the contraindications of MOVFOR® 200MG?
- Hypersensitivity to molnupiravir or any other component of the drug molnupiravir
- Pregnancy or pregnancy planning
- Breastfeeding period
- Children under 18 years of age
What are the Side effects of MOVFOR® 200MG?
The most frequent adverse reactions reported during treatment with MOVFOR® Molnupiravir at a dose of 800 mg every 12 hours for 5 days and for 14 days after taking the last dose of the drug were diarrhea (3%), nausea (2%), dizziness (1%) and headache (1%), which had mild or moderate severity.
Contents
- 1 Buy MOVFOR® 200MG Capsules Online
- 1.1 Order MOVFOR® 200 MG Molnupiravir Capsules
- 1.2 What do we know about its effectiveness MOVFOR® 200 MG Molnupiravir Capsules?
- 1.3 Molnupiravir is embedded in the coronavirus genome and causes it to mutate to death
- 1.4 How to take MOVFOR® 200 MG Molnupiravir Capsules?
- 1.5 What is the dosage for MOVFOR® 200 MG?
- 1.6 Special patient groups for the drug MOVFOR® 200 MG:
- 1.7 What are the contraindications of MOVFOR® 200MG?
- 1.8 What are the Side effects of MOVFOR® 200MG?